Updated Guidelines To Monitor Metal on Metal Devices by UK’s MHRA 2017
Original MEDICAL DEVICE ALERT by UK MHRA 2017 Available Here
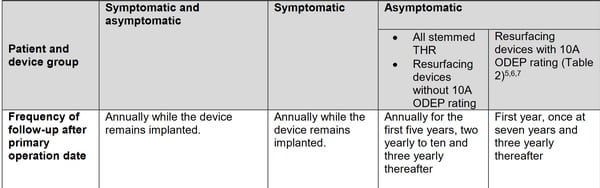
The Medicines and Healthcare products Regulatory Agency (MHRA) UK has called for longer-term monitoring of all patients fitted with metal-on-metal hip implants, as a precautionary measure, in an update to its original guidance issued in 2012.
“MHRA, in colsultation with its independent Metal-on-Metal Expert Advisory Group (MoM EAG), has continued to monitor the performance of MoM hip joint articulations for the occurrence of soft tissue reaction associated with these devices. However, some patients will develop progressive soft tissue reactions to the wear debris associated with MoM articulations.”
Advice varies according to device, age and sex of patients. The BHR is device with a 10A ODEP rating. Those patients are recommended to have follow-ups and blood tests: First year, once at seven years and three yearly thereafter.