Hip Resurfacing Devices with Poor Performance Records
I find it important to provide the most reliable information available on the
Surface Hippy Website. Since it started in 2005, there have been a variety of
hip resurfacing devices available. Only three have been approved by the
FDA. The information posted on Surface Hippy is based on National Registry
Information, Medical Studies and Surgeon Articles.
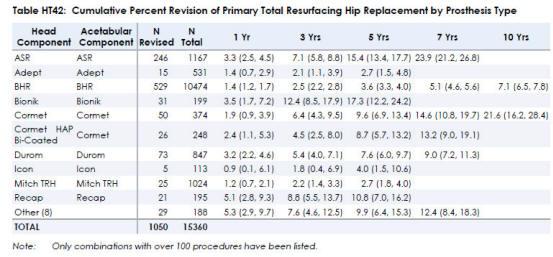
Unfortunately, some hip resurfacing devices have not provided good outcomes
to patients and have been removed from the market. As hip resurfacing
devices continue to be used, there is now a longer history of outcomes available
up to 10 years reported in the Australian Orthopaedic Association National Joint
Replacement Registry 2013. The ASR was removed from the market. The Corin
Cormet had very poor results according to the Australian 2013 National Registry.
I decided to no longer provide detailed information about devices
that had poor outcomes and the surgeons using them. That decision
does not indicate that a patient won’t have an excellent outcome with those
devices and that they can not have a successful surgery by surgeons choosing to
use those devices. That decision means that I want to help people learn
about hip resurfacing and excellent outcomes. I want to provide detailed
information about successful hip resurfacing devices and the surgeons that
choose to use them.
Information from Australian Orthopaedic Association National Joint
Replacement Registry 2013
Full Reports Available: