Informed Consent for HRA Thomas P. Gross MD Updated 3/12/2014
Updated 3/12/2014
Original PDF Here:
https://surfacehippy.info/pdf/dr-gross-HRA consent-3-12-2014.pdf
Dr. Gross has now performed over 3500 Hip Resurfacing Arthroplasty (HRA)
procedures over the last 13 years. Most failures occur during the first year of
the healing period. However, there is a slow rate of failure that occurs over
time. Therefore the overall failure rate increases for a group of patients as
the length of follow-up increases. In the Journal of Arthroplasty 2011, we
reported that our Corin Hybrid HRA achieved a 93% survivorship at 11 years
follow-up. Longer-term data is not available. The first 1000 Biomet uncemented
HRA were reported in the Journal of Arthroplasty achieving an overall 6-year
survivorship of 98% (99% for men and 96% for women) Not all complications lead
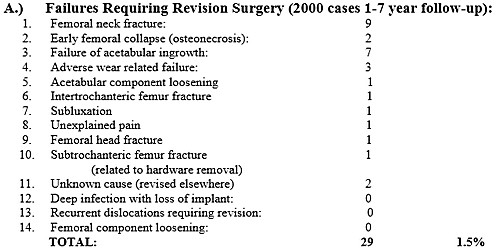
to failure. Below is a complete list of major complications (not just failures)
in the first 2000 uncemented HRA using the Biomet uncemented system:


D.) Hip Resurfacing Survivorship
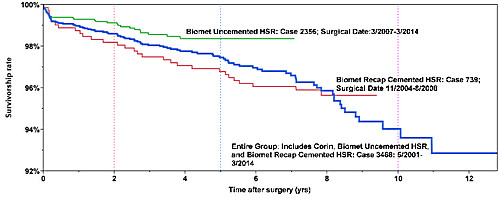
3468 cases over 13 years
Survivorship of hip resurfacing continues to improve as we gain more
experience and find measures to prevent failures. Theses survivorship curves
give the reader an opportunity to see what the odds are that their implant will
still be functioning at some time point after implantation. We have used three
implant systems in the last 13 years. We present three Kaplan-Meier Survivorship
curves: the best are uncemented Biomet devices (Green), the second are all of my
hip resurfacing combined (blue), and finally hybrid fixation Biomet devices
(Red). The earliest Corin Hybrid group is not listed separately.

E.) Reducing Failures in HRA:
In the last 10 years we have learned what patients with particular
characteristics are at higher risk for failure. The strategy of many experts is
patient selection. If they avoid performing hip resurfacing on higher risk
patients, their overall results will improve. I disagree with this defeatist
strategy…and no one has demonstrated that these high-risk patients will fare
better with a THR.
My improving results are not achieved by patient selection. I have always
practiced minimal patient selection and my criteria have not changed much over
the last 13 years. Instead, my goal is to find treatment modifications to
improve the results in patients that are traditionally identified as higher
risk. In some cases, we have already accumulated scientific evidence of improved
results; in other cases, we still need more time to gather data to be certain
that our treatment modifications have improved results. Examples of our
innovations include:
1. Femoral neck fracture: stratifying risk of femoral neck fracture by bone
density and BMI and treating higher risk patients with slower weight bearing and
bisphosphonate drugs. We have demonstrated that this substantially reduces risk.
Fracture risk now less than 0.07%.
2. Failure of acetabular implant attachment: dysplasia patients are at higher
risk because of socket deformities. Use of Trispike acetabular components in
severely deficient sockets has eliminated these failures in this high-risk group
since 2007.
3. Femoral cysts: Bone grafting cysts instead of filling them with cement has
resulted in eliminating femoral cysts as a risk factor for failure in our
patients.
4. Femoral Loosening: The major source of late failure in my cemented femoral
components. We have demonstrated that uncemented femoral components are at least
as good as cemented ones at up to seven years follow-up. We have not yet had one
case of femoral loosening after 1 year in over 2500 cases. We have had no
femoral failures in high-risk osteonecrosis cases (others avoid resurfacing
these).
5.Adverse wear related failure (AWRF): We have determined that this is caused
by faulty implant design and certain acetabular component positions,
particularly high acetabular inclination angles (AIA). We have published a
robust guideline for component positioning and an intraoperative XR technique
that allows us to achieve this goal in over 99% of cases. The last AWRF case was
created in 6/2009 and has been revised with an excellent final outcome.
6.We have published a report demonstrating that revision of failed HRA can be
performed with a high success rate (96% 6-year survivorship) nearly as good as
primary surgery. Unlike other reports, our success rate for revising for AWRF
has been 100% without major complications. The strategy used was repositioning a
new metal bearing in the correct position
not using the failed strategy of changing to small plastic bearings.
Past results do not guarantee future complication rates. Although the above
represent the most common complications associated with this procedure, others
could also occur. We continue to strive to make improvements, and hope that
these complication rates can be further decreased as we gain even more
experience.
- Dr. Gross is the operating surgeon (No trainee will perform your
operation). - Dr. Gross developed the Biomet implants but no longer receives royalties
for these implants. - Biomet Recap and Magnum components are FDA approved. Use as a total hip
resurfacing is however considered off-label. - Information from your treatment is used for research purposes, but you
will not be identified.
If you have any questions about the above information, please don’t hesitate
to ask.
I have reviewed the above and understand the risks involved with this
operation. I would like Dr. Thomas Gross to perform hip resurfacing on me.
_________________________________________________ _____________
Patient Signature
Date
_________________________________________________ ______________
Witness
Date
