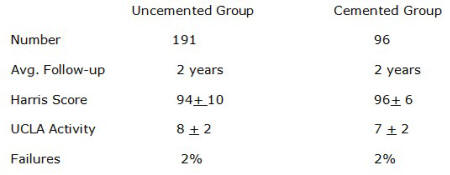|
Midlands Orthpaedics p.a.
Thomas P. Gross, M.D.
Current status of uncemented femoral components in hip resurfacing.
January, 2010
Uncemented femoral components for metal on metal total hip resurfacing have shown excellent results during the initial 3 years that I have been using this new technology. The early results that I have achieved in 0ver 800 cases since March 2007 are equivalent to the early results that I achieved with the same brand cemented femoral
component.
Uncemented fixation of implants to bone is a proven technology that has
generally surpassed the durability of cemented fixation to bone in traditional
hip replacement surgery. In the long term (at 10 years) a higher percentage of
hip implants using uncemented fixation still remain attached to the bone than
cemented implants, especially in younger more active patients.
Fixation of total hip implants to bone can be accomplished by cement or by
porous ingrowth technology (uncemented). Cement fixation is immediate. Cement is
an acrylic material (methylmethacrylate) that is very brittle and also fairly
toxic to bone cells. Cemented implants gradually loosen from the bone over time
by reaction to the cement itself and due to gradual fatigue failure of this
material. This process is faster in more active patients and faster in implant
situations where the cement is stressed by shear forces rather than by
compression forces.
Uncemented components are initially held to the bone by a very tight press-fit
which is achieved by accurately preparing the bone so that the implant can be
tightly hammered-on. The implants are so tightly wedged–on that the patient can
bear full weight on them immediately. They do require a period of six to twelve
months of bone ingrowth before they are considered well fixed. There is usually
a small chance of failure of this bone ingrowth process in uncemented implants
(<1%). But if ingrowth occurs, it is much more durable than cemented fixation
and rarely fails in the long term.
Because orthopedic surgeons in America have come to a consensus on the
superiority of uncemented fixation in total hips, uncemented fixation has
virtually completely replaced cemented fixation in stemmed total hip
replacements, despite the fact that these implants are more expensive. 99% of
acetabular (socket) components that are used today are of the uncemented type,
as are about 90% of femoral stems.
In hip resurfacing there is universal agreement that uncemented fixation is
superior for the acetabular component. However, until recently, uncemented
femoral components have not been available, therefore most hip resurfacing
operations in the past have employed cemented fixation of the femoral component.
At the time that I began hip resurfacing in 1999, there was not yet general
agreement that uncemented fixation was superior to cement in hip replacements.
However, the evidence was mounting that uncemented fixation was better. I
therefore did not think it was logical to use cemented fixation in hip
resurfacing, an operation developed specifically with the more active younger
patient in mind. The only companies pursuing hip resurfacing at the time were
two small English companies: Corin and Midland Medical Technology (maker of the
Birmingham implant). I suspect that they did not have the financial resources to
develop a more complicated uncemented femoral component with the precision
instrumentation required at that time. I originally proposed an uncemented
femoral component to Corin 10 years ago, but they were unable to manufacture it
at that time.
I therefore worked with Biomet on an uncemented femoral component and the
precision instrumentation required for this implant for five years. I first
began implanting it in March 2007. The Biomet component has a full coating of
Titanium plasma spray under the entire under-surface of the femoral component.
Recently we have added an additional layer of hydroxylappatite (HA) to increase
the speed and extent of bone ingrowth. This is the best implant available to
maximize the chance of bone ingrowth. When I started working with Biomet to
develop an uncemented femoral component, Corin also started to work on one. They
were able to bring it to market in Europe first; however, their component is
only partially porous-coated (less than 50%) with Titanium (but it does have
complete hydroxyl appetite coating). I personally do not believe this is good
enough for long-term fixation (>10 years), but nobody knows for sure yet. It is
not yet available in the US.
Theoretically, cement is the weak link when long-term (> 10 years) fixation of
the femoral component is contemplated. If uncemented femoral components can be
shown to achieve reliably high rates of ingrowth in the short term, they will
probably outperform cemented femoral components in the long-term.
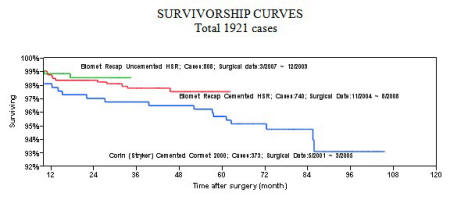
At this point with nearly 2-year follow-up data on a matched group of patients,
I see no difference in results whether cement or uncemented fixation is used. At
this point we can be fairly certain that bone ingrowth has occurred in these
components. Except for two cases where osteonecrosis occurred in the femoral
head at 1 year, we have had no failures of bone ingrowth in 430 patients that
have are at least one year postop, and 191 that are at least two years postop.
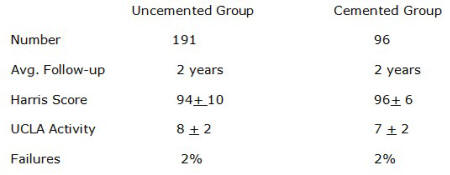
In summary:
- Uncemented
femoral resurfacing components are now available from BIOMET for any patient
who desires them.
- No other
companies are yet selling these in the US
- Corin has
had an uncemented femoral component available in Europe for several years.
- At 2 years
of follow-up there is no difference in the failure rate between cemented or
uncemented femoral component.
- Uncemented
fixation of implants is more durable at 10 years than cement in hip
replacement surgery especially in young active patients.
- Most
clinical data on hip surface replacement to date is based on an uncemented
acetabular component and a cemented femoral component.
- I now use
uncemented components on virtually all hip resurfacing operations, unless
the patient specifically requests the cemented femoral device.
Thomas P. Gross, MD
1/10/2010
|